|
The range and specific energy loss of positrons is about the same as that of negative beta particles, but they are different in that they annihilate with an electron from the absorbing material at the end of their track, yielding two 0.511 MeV photons. During the decay event a proton converts to a neutron and a positive electron, or positron, which is ejected from the nucleus. It has the same mass as an electron, but carries a positive charge. A positron, the anti-particle of a beta particle, is emitted by a proton-rich nucleus. For example, the beta dose rate at 3 cm from a 1 mCi vial of P‐32 is:Ī few isotopes, such as 11C, 13N, and 18F, decay by positron emission. Where X is the dose rate measured in rad/hr, A is activity in Ci, and r is distance in cm. The dose rate from a point beta source with energy greater than 0.5 MeV is: A beta emitter can also cause radiation dose if ingested.Ī low atomic number material such as plastic is used for shielding a beta emitter. They pose an external source of radiation dose to the skin and eyes. Properties of β‐ particlesĪs with alpha particles, beta particles are completely attenuated by small thicknesses of common materials. Neutrinos have little mass and no charge, and do not frequently interact with matter. To explain this, Pauli postulated the existence of a new particle, the neutrino (ν), emitted simultaneously and sharing the energy of the decay event with the beta particle. For a low energy beta particle, we might ask where the missing energy has gone. The maximum and average are characteristic for the isotope. Unlike the discrete energies observed for alpha particles and gamma rays, the average kinetic energy of all beta particles from a given isotopic sample is about one‐third the maximum energy that is possible for that isotope. See Table 1.1 for other examples of beta emitters. The higher energy particles are more penetrating. The maximum kinetic energy of the beta particle, in this example 0.156 MeV, can range from as low as 0.019 MeV for a 3H decay to as high as 1.7 MeV for a 32P decay, or 3.3 MeV for a 214Bi decay. During the radioactive decay event, the neutron reverts to a proton, an energetic electron and a neutrino that escapes the nucleus. This leads us to observe that the neutron is essentially a proton with an attached electron. It differs from an electron only because it is a product of radioactive decay. The beta particle is an electron that has been ejected from a neutron‐rich nucleus. Ingestion of radium can cause a very large radiation dose to blood‐ forming cells. For example, radium is in the same column of the periodic table of elements as calcium, and is a bone seeker. 
However, if ingested, they can deliver a very large radiation dose to tissue. Therefore, alpha particles pose no external hazard. A thin piece of paper, or the layer of dead cells on your skin surface, will completely attenuate a beam of alpha particles. Thus, they lose all of their kinetic, ionizing energy after travelling a very short distance in any medium. Properties of α‐ particlesīecause of their +2 charge and relatively low velocity, alpha particles are densely ionizing, depositing an enormous amount of energy at each collision with an attenuating atom. Almost all radionuclides that decay by alpha emission have atomic number greater than 83 (bismuth). They are frequently accompanied by high energy gamma rays. Most have an initial kinetic energy of about 5 MeV. For example,Įach alpha particle has a charge of +2 and a mass of 4. It is associated with the radioactive decay of elements of high atomic number. The alpha particle is simply a helium nucleus, comprised of two protons and two neutrons. For a more complete introductory discussion, see Alpen ch. Decay schemes for selected isotopes also appear in many texts and reference books. One of the best sources of information on decay modes is the Chart of Nuclides published by the Knolls Atomic Power Laboratory. There are a variety of radioactive decay modes. 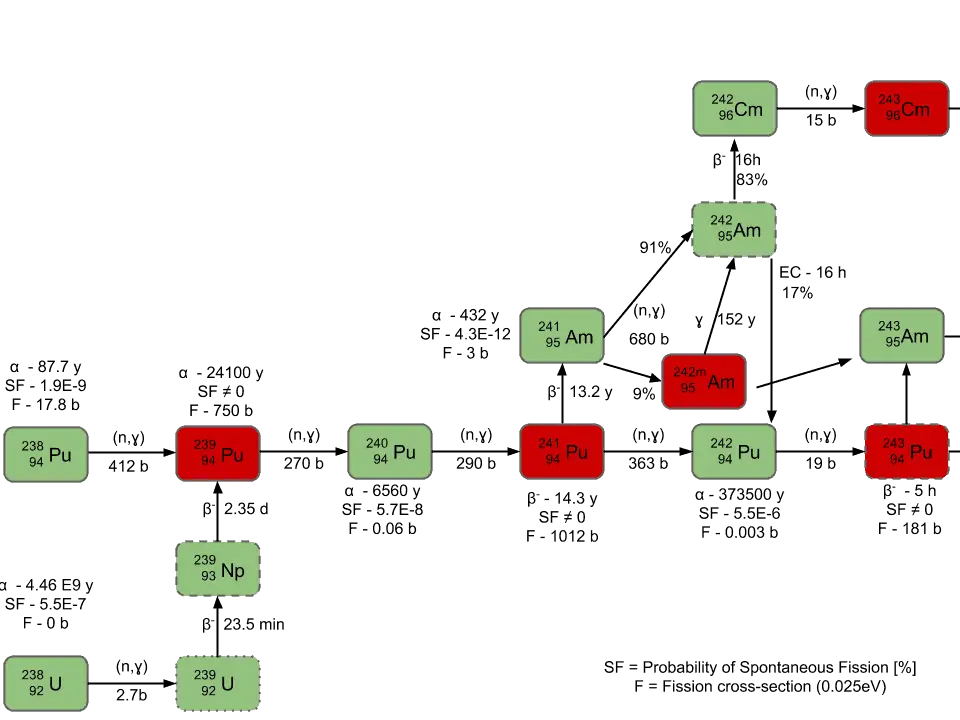
Each decay event involves loss of mass or charge. This event is called a nuclear transformation, a decay, or a disintegration. Radioactivity is the spontaneous emission of charged particles or photons by an atomic nucleus that is in an unstable configuration.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
